- Blog
- Sonic mania game gear mods
- Ps3 eye camera driver download
- Grateful dead dancing bear rings
- Example of monthly business expenses excel spreadsheets
- Ghost of tsushima map full
- Unity funeral home cremation
- Download missing media driver for windows 10
- Notion weight loss tracker template
- Noteburner apple music converter for mac
- Water density kgl
- Atlanta ga time zone gmt
- The periodic table printable high school chemistry
- Alligator clips hobby lobby
- Solitaire tripeaks free coins android
- Cover letter examples healthcare
- Rocksmith no cable patch 2014
- Hotkey script for das trader
- Space shuttle endeavour crew
- Easy vanilla fudge recipe karo syrup
- Grounds for a motion to dismiss
- Windows 10 stuck on lock screen
- Best family movies on netflix 2020
- Herchey script font free download
- Basic lung sound
- What is the best background eraser app
- Paw patrol graphic svg free
- Exe barney on pubg
- Sports head basketball unblocked b games
- Sony vegas pro 14 serial number free
- Total war rome 2 empire divided factions
- Adobe after effects cs6 download torrent
- Free microsoft word calendar template
- Landscape rawtherapee presets
- Minions butt wallpaper
- Sakura dungeon installing patch breaks game
- Asd definition dsm 5
- Iomega x1de usb driver windows 7 download
- Farming simulator 22 indian mod download
- Brochure design templates free download for word
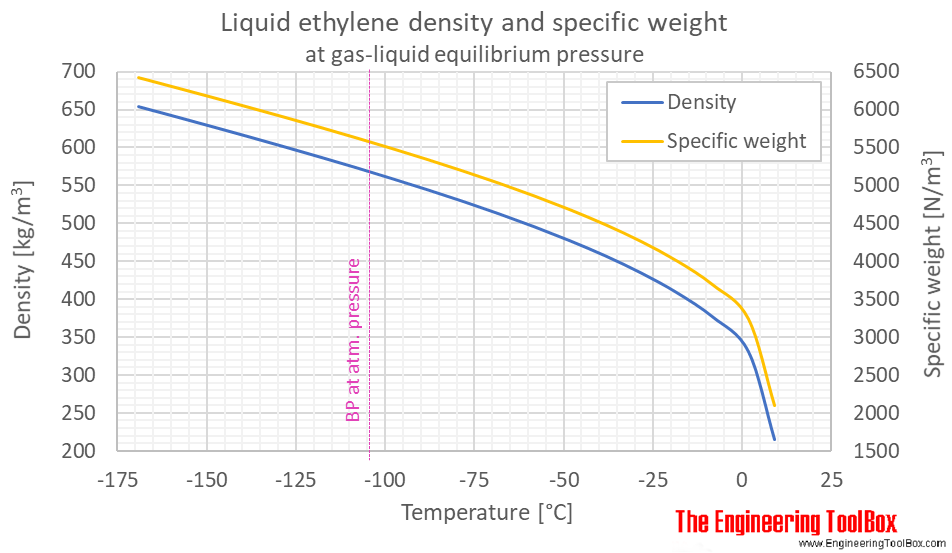
Phase diagram included.Ĭritical Temperatures and Pressures for some Common SubstancesĬritical temperatures and pressures for some common substances like air, alcohol, ether, oxygen and more.ĭensity vs. Introduction to - and definition of - Bulk Modulus Elasticity commonly used to characterize the compressibility of fluids.Ĭarbon Dioxide - Thermophysical PropertiesĬhemical, physical and thermal properties of carbon dioxide. Phase diagram included.Ĭhemical, physical and thermal properties of benzene, also called benzol. Thermal properties of air at different temperatures - density, viscosity, critical temperature and pressure, triple point, enthalpi and entropi, thermal conductivity and diffusivity and more.Ĭhemical, Physical and Thermal Properties of Ammonia. Hot and cold water service systems - design properties, capacities, sizing and more.Ĭhemical, physical and thermal properties of acetone, also called 2-propanone, dimethyl ketone and pyroacetic acid.
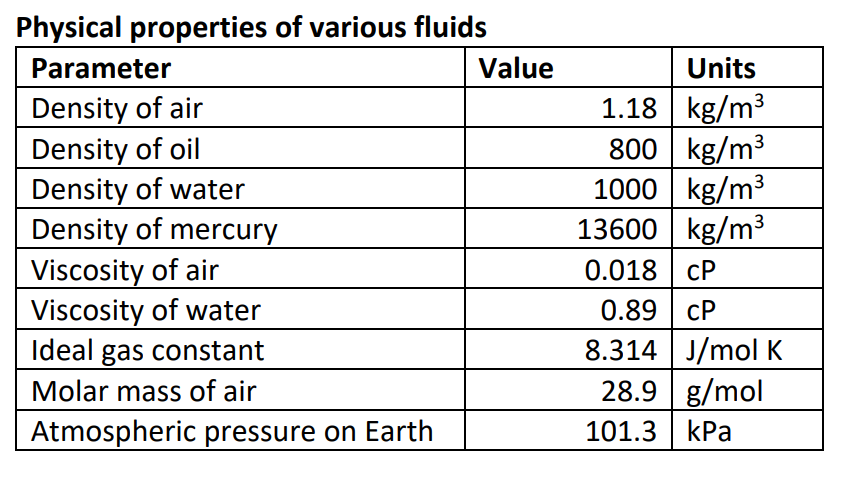
Steam & condensate systems- properties, capacities, pipe sizing, systems configuration and more. Material properties of gases, fluids and solids - densities, specific heats, viscosities and more. See also more about atmospheric pressure, and STP - Standard Temperature and Pressure & NTP - Normal Temperature and Pressure ,Īs well as Thermophysical properties of: Acetone, Acetylene, Air, Ammonia, Argon, Benzene, Butane, Carbon dioxide, Carbon monoxide, Ethane, Ethanol, Ethylene, Helium, Hydrogen, Hydrogen sulfide, Methane, Methanol, Nitrogen, Oxygen, Pentane, Propane, Toluene and Water.
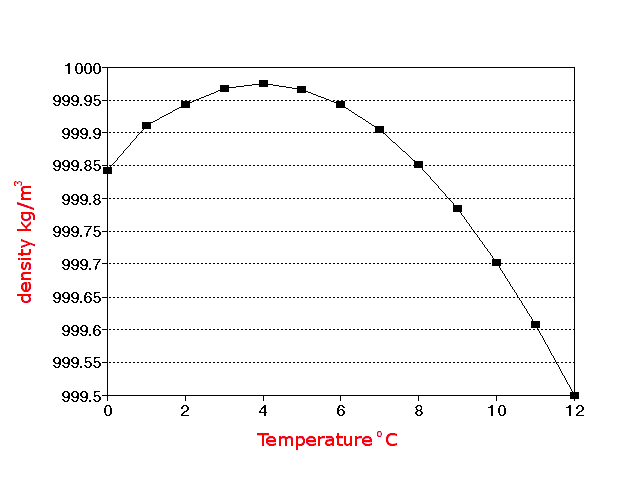
Ionization Constant, pK w, of normal and heavy water with varying temperature.

Thermodynamic properties of heavy water - D 2O: Heavy water ( deuterium oxide, 2H 2O, D 2O ) is a form of water that contains a larger than normal amount of the hydrogen isotope deuterium (= heavy hydrogen = 2H = D), rather than the common hydrogen-1 isotope ( 1 H = H = protium) that makes up most of the hydrogen in normal water.
- Blog
- Sonic mania game gear mods
- Ps3 eye camera driver download
- Grateful dead dancing bear rings
- Example of monthly business expenses excel spreadsheets
- Ghost of tsushima map full
- Unity funeral home cremation
- Download missing media driver for windows 10
- Notion weight loss tracker template
- Noteburner apple music converter for mac
- Water density kgl
- Atlanta ga time zone gmt
- The periodic table printable high school chemistry
- Alligator clips hobby lobby
- Solitaire tripeaks free coins android
- Cover letter examples healthcare
- Rocksmith no cable patch 2014
- Hotkey script for das trader
- Space shuttle endeavour crew
- Easy vanilla fudge recipe karo syrup
- Grounds for a motion to dismiss
- Windows 10 stuck on lock screen
- Best family movies on netflix 2020
- Herchey script font free download
- Basic lung sound
- What is the best background eraser app
- Paw patrol graphic svg free
- Exe barney on pubg
- Sports head basketball unblocked b games
- Sony vegas pro 14 serial number free
- Total war rome 2 empire divided factions
- Adobe after effects cs6 download torrent
- Free microsoft word calendar template
- Landscape rawtherapee presets
- Minions butt wallpaper
- Sakura dungeon installing patch breaks game
- Asd definition dsm 5
- Iomega x1de usb driver windows 7 download
- Farming simulator 22 indian mod download
- Brochure design templates free download for word
